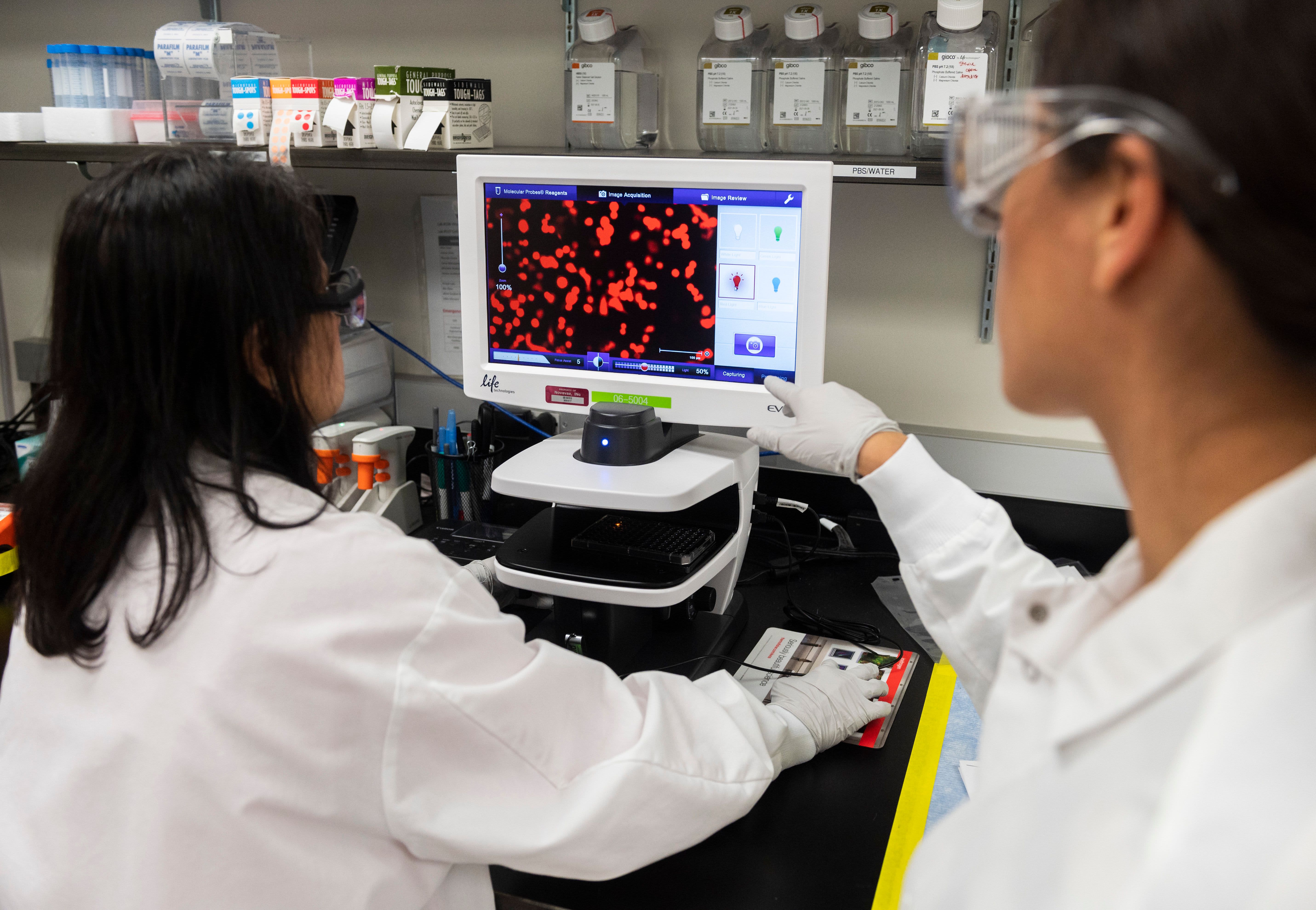
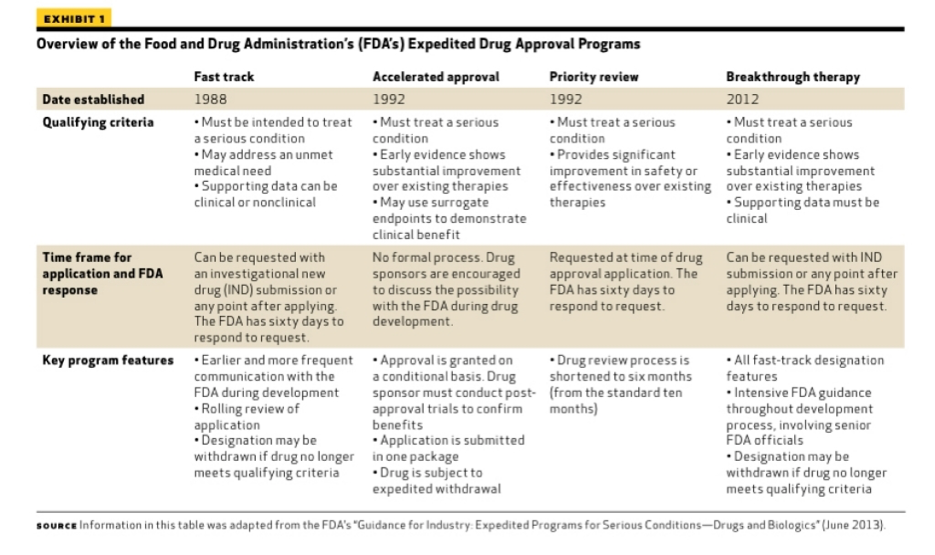
Avantor supports fast-tracking of clinical trials related to COVID-19 disease progression with direct-to-patient custom kitting solution | Avantor

Breakthrough Drug is a Breath of Fresh Air for Cystic Fibrosis Patients — Journal of Young Investigators

Fast track MHRA approval for Vicore Pharma's COVID-19 clinical trial received in record time – orphan reach™- A Rare Disease CRO for Orphan Drugs

A First Successful Pilot Project for CATALIS' New FAST TRACK Evaluation Service: 4 Quebec Institutions Ranked in the Top 5 of the World's Fastest Sites - CATALIS